Meet the Measurements
Familiarize yourself with the universal constants with these hands-on laboratory experiments.
Teacher Tips!
Many activities have a teacher view and a student view, and teachers can switch between those views by clicking the blue button in the upper-right. Students will not see this option - only teacher accounts see both views. The teacher view will start with overview text, if available, to frame the activity and get you started. This view will also have teacher tips and suggested answers to student questions spread throughout the activity. The teacher text interleaved with student-facing text will be in italics and should appear as a different color on your screen. Teacher tips are designed to help you deliver a learning experience that is best suited for your classroom.
Printing Reminder
Whichever view you see on your screen is what will print. You can print this activity without teacher tips by selecting the student view, or print with teacher tips by switching to teacher view. Simply use the standard print function available for your web browser. No extra steps are required.
Title of Activity:
Meet the Measurements
Curriculum Collection:
NIST: Universal Constants, Lesson 1
Description:
In this activity, students explore the basis for SI measurements, beginning to consider issues of accuracy and precision that they will examine further in later activities.
Target Grade Level:
8-12
Discipline or Course:
Physical Science
Time Frame:
Approximately 2 class periods
Suggested Grouping:
3-4 Students
Key Vocabulary:
- Measurement
- Accuracy
- Precision
- Constant
- SI
Teacher Prep:
This is designed as a station activity through which students rotate.
- Enlarge the images for Station 2. Find as many measurement tools as you can for comparison.
- For Station 3, small diet scales can work.
- The paper template measuring tool in the last station must be reproduced exactly. A centimeter measuring tape can also be substituted, but there will be a lack of precision as it is bent around the hand and fingers.
Differentiation:
- Where mathematics is used, a guide to lead students through the calculations is helpful.
- Two definitions are included at each station: accuracy and precision. Do not expect students to completely understand the distinction yet. Further definitions can be found in the glossary.
Taking It Further:
- Students can suggest measurement challenges in their own environment and match the standards to the processes.
- Students can discuss the measurement of the size of molecules, particles (moles), and the measurement of time-related to chemical reactions.
Suggestions for Remote Learning:
- While all of the stations could be replicated at home, Station 2's Slinky, Station 3's balance, and the lights in Station 5 would probably require purchase.
- A normal eyedropper can be used instead of a graduated pipette. If you let a drop fall "of its own weight" there will be very close to 20 drops/mL. This can be used to estimate the quantities for the activity.
STUDENT CONTENT BELOW
When we describe a phenomenon, it's common to ask: How big? How small? How much? To communicate in the most scientific way, we need to ask another question: How do we know for sure?
Materials (per Station)
Station 1: Poisoned Frogs
-
- 100 mL beaker
- Graduated pipette or eye dropper
- Iodine or biological stain
- Water
- (Optional: Colorimeter or mobile app for measuring - many are available for free on most phones)
Station 2: A Wrinkle in Time
-
- Slinky™
- Phone camera with "burst" function and timer
Station 3: Precise Prescriptions
-
-
Aspirins ("baby" ~ 80 mg), at least 2 brands, 10 each
-
Electronic balance / scale
-
Station 4: Get Your Bearings
-
- Distance meter (carpenter's laser tool, or mobile app for measuring; many are available for free on most phones)
- Trundle wheel
- Ruler
- Tape measure
- Meter stick
- Community map
Station 5: Starlight, Star Bright
-
- Batteries in holder
- Decorative string lights
- Light meter (device, or mobile app for measuring; many are available for free on most phones)
- Box (like a shoe box)
- Flashlight
- Wiring
- NASA space images
- Cutting tool
Station 6: Physical Therapy
-
- Paper template of measuring tool
Safety Precautions:
- Station 1: Eye protection is needed for each student. Some people are allergic to iodine. If you are allergic, use a biological stain instead. If allergy status is unknown, use gloves and be very cautious.
- Station 3: Use all appropriate caution when handling aspirin.
- Station 5: Use eye protection when cutting.
Station 1: Poisoned Frogs
In a controversial study, Stanford University biologist Tyrone Hayes found that incredibly small concentrations of the weed killer atrazine could inhibit the development of frogs. Atrazine is in many weed-killing products. Its molecular structure is similar to natural hormones, and so the body stops making its own hormones. Hayes found that as little as 0.1 part per billion of the chemical can inhibit sexual development in male frogs. Environmental tests show that farm runoff often carries a concentration of as much as 40 parts per million atrazine.
Procedure:
- Fill a beaker with 100 mL of water at room temperature. You might place the beaker on a white piece of paper or background to help you see changes in color more easily. Your challenge is to determine the minimum concentration of solute (a dye or tincture) that will produce a change in the color that you can sense/see. You will add the dye 1/20 mL at a time. That is about one drop. If you do not have a pipette that can add the solute with that precision, allow drops to fall of their own weight.
Solvent (Water) mL
Solute Drops
mL
Parts per million
100
100
100
100
100
100
- Think first: How many of your beakers would equal 1 million mL? 10,000
- Now add solute 1/20 mL at a time. Stir gently. Check for a color change. Record the minimum concentration of dye that results in a color change that you can see.
Think:
- What is the greatest precision you can get with your dropper or pipette? These answers will vary.
- What is the smallest concentration you can sense? These answers will vary.
- If you used an electronic instrument (colorimeter or mobile app), how much more precision did you achieve? These answers will vary.
- Is a visual observation of a color change accurate? Precise? It could be accurate but not precise.
- What procedure or instrument could you use to make your analysis more precise? A colorimeter or light meter can make the observation more precise.
- Now imagine a small pond shaped like half a sphere, with a depth (radius) of 10 m.
- What is the volume of water in the half-sphere pond? 2,094 cubic meters
- How much atrazine would be needed to pollute to 0.1 ppb? 2-9.4 milliliters
Constant:
In chemistry, concentration is normally measured in moles. A mole used to be defined as the amount of a substance that contains as many particles as there are atoms in 0.012 kilograms of carbon. Now, the Avogadro constant defines the number of particles in a mole. Avogadro's number of electrons equals one mole of electrons. Similarly, Avogadro's number of water molecules equals one mole of water. In this revised SI, NA equals 6.02214076 x 1023 particles per mole.
Vocabulary:
- Accuracy: The degree to which the results of a measurement conform to the correct standard
- Precision: The quality of being exact; the reproducibility or repeatability of a measurement.
Station 2: A Wrinkle in Time
Look carefully at the first photo. It was taken by Louis Daguerre in 1839 in Paris.
He had to leave his lens open 7 minutes in order to get the picture. In the lower left, you will see the faint image of two people who stood still long enough to be caught on the picture — the first picture of human beings ever made. It took time!
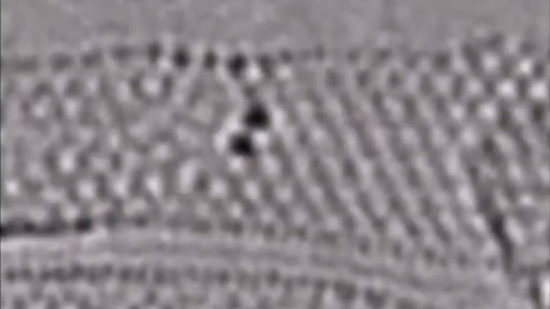
Now look at the second picture; the image looks blurry, but it shows something remarkable.
For the first time in 2019, scientists at the University of Nottingham were able to capture two atoms forming a molecule (dirhenium). The event occurred in 0.71 seconds.
Procedure:
In this activity, you'll experiment with the burst function of your phone camera, to try to capture one wave cycle of a Slinky™.
- Clamp one end of the Slinky™ firmly to a table.
- Make a transverse wave by starting a force at one end of the Slinky™.
- Ask a partner to take photos of the wave movement at various pulse rates.
- What shutter speed allows you to capture just one compression of the wave? These answers will vary depending on the speed of the wave the students are creating.
Think:
- What is the greatest precision you can get with your burst function? That's the inverse of shots per second. These answers will vary.
- How does the image change if the interval is too short? You only get an image of a piece of the wave.
- How does the image change if your interval is too long? You get several cycles.
- Is a visual observation precise? No, everyone's sense of time differs. Realize that the scientific definition of “precise” needs more explanation for students at this point.
- How does time affect the way that we understand natural phenomena? These answers will vary.
- Give an example of something that occurs faster than we can see. Movements of a hummingbird, flickering of pixels on a television screen.
- What phenomena could be measured more precisely if shutter speed were faster? Examples like those above might be repeated here. Any chemical reaction is also a good response.
Constant:
The constant used for time (second) is based on the frequency of radioactive discharge (ticks) of a cesium-133 atom clock: 9,192,631,770 Hz per second.
Vocabulary:
- Accuracy: The degree to which the results of a measurement conform to the correct standard
- Precision: The quality of being exact; the reproducibility or repeatability of a measurement
Station 3: Precise Prescriptions
In formulating pharmaceuticals, very precise measurements are vital. In some cases, even 0.01 mg might make the difference between a cure and a negative reaction. This is especially true of hormones and a new class of drugs called monoclonal antibodies. When a manufacturing process does not produce a highly precise formulation every time, the consequence is often a complex and expensive recall. In the worst case, it might even result in injury to patients and legal action against the company. In addition to precision in the active ingredient of the drug, the manufacturer must ensure precision in the other components of the pill or capsule. The coating and/or filling material of a drug may affect how it is absorbed into the bloodstream.
A "baby aspirin" contains a dose of 80 or 81 mg of acetylsalicylic acid with a coating that is often flavored. Many adults take "baby aspirin" as a long-term treatment to reduce the likelihood of stroke or heart attack. Each of the many brands has a slightly different formulation of all the ingredients except the aspirin.
Safety Warning: Even though the dose is very small, treat this as medicine with all caution.
Procedure:
- Collect a sample of 10 tablets from each bottle (brand) of aspirin at random.
- Mass all 10 together.
- Determine the average mass for 1 tablet of each brand.
- Subtract 80 mg or 81 mg (the mass of the active ingredient on the label) and fill in the last column of the chart. Repeat the procedure to see if your sample is consistent.
Brand |
Mass of 10 Tablets |
Average Mass of 1 tablet |
Mass of active ingredient |
Mass of other ingredients |
Percent active ingredient |
Average percent active ingredient |
|---|---|---|---|---|---|---|
Think:
- Compare the mass you got for 10 tablets of a specific brand in your first and second trials. Then compare it to the measurements from other groups. Are all the measurements consistent? Within the range of precision of most inexpensive balances, inconsistencies may or may not be detectable.
- If you had a more precise balance that gave you more significant figures (decimal places) could your answer to question 1 be different? It may be different.
- What is the greatest precision your balance can achieve? These answers will vary. If you have two balances with different levels of precision, compare the results.
- Could your balance give you a more precise measurement if you massed only one tablet at a time? Probably not.
- How would a pharmaceutical manufacturer increase precision? Better balances, more specific procedures.
Constant:
The SI unit of mass is the kilogram. Metrologists once measured mass by comparing objects to a standard object, or artifact. Now they evaluate the energy equivalent of the mass in terms of Planck's constant, h, which is equal to exactly 6.62607015 × 10-34 Joule seconds.
Vocabulary:
- Accuracy: The degree to which the results of a measurement conform to the correct standard
- Precision: The quality of being exact; the reproducibility or repeatability of a measurement
Station 4: Get Your Bearings
Have you ever been lost? 2,000 years ago in Mesoamerica, evidently the Olmec people got lost, too! Scattered throughout the dense jungles where they lived are statues that archaeologists call "fat boys" or "potbelly sculptures." At first they thought they were religious artifacts, but now we know they had a surprisingly advanced function. Their heads, and sometimes other body parts, are magnetic, so jungle travelers could use them to guide their compasses. The Olmecs measured distance from statue to statue.
How would you get your bearings?
Begin describing your location as precisely as possible as you would explain it to:
- A friend at the door trying to find you.
- A delivery person trying to bring you a bouquet of flowers from downtown.
- An alien in a UFO trying to abduct you.
Discuss with your group: How are these directions different? Which must be most precise?
Procedure:
- Find several different measurement instruments. You can also download a mobile app for measuring; many are available for free on most phones, and you can use your phone's camera as a measuring instrument. Compare the precision of each instrument. It's reasonable to estimate one unit of measurement between the marks; if a meter stick is marked in centimeters, you might read to 0.5 centimeters.
Instrument
Smallest gradation
The smallest measurement you can estimate
Precision
- Now use the tools to measure the distance from your seat to the wall. Think about the percent of each measurement that is uncertain. For example, if the distance was 3 meters and the tool was marked in centimeters, then the uncertainty would be 0.5/300 or 0.16 percent.
- Go a bit further. Find the distance from the room you are in to the front door of your school's central administration office. Use a map or a GPS app on a phone to determine the distance. Remember that if you ask GPS for driving distance, it follows streets. You need to do that, too. Calculating "as the crow flies" (in a direct line, regardless of streets or other topographical features) is a different problem.
Think:
- How do you know that a measurement is accurate? What standard would have been used when your tools were designed? It's likely that the physical kilogram artifact was the standard.
- What determines whether your measurements are precise? The way the tool is marked and your procedure in using it.
- How could precision be improved? Better tools, better procedures.
- GPS is a system based on the time it takes for a signal to go from an instrument to satellites and back again. What are the factors that would determine the accuracy of a GPS measurement? Clock, the transmission of signal, software.
Constant:
The SI unit of distance is the meter, based on c, the speed of light. c is equal to 299,792,458 meters per second. Light speed is always constant in a vacuum, so you can define any distance by the time it would take for light to travel that far.
Vocabulary:
- Accuracy: The degree to which the results of a measurement conform to the correct standard
- Precision: The quality of being exact; the reproducibility or repeatability of a measurement
Station 5: Starlight, Star Bright
Ancient traders were guided by the stars, traveling overland and across oceans. Today it might be a lot harder to see those same stars. The light we produce for our Earth-bound activities confuses our vision and prevents us from having clear views of most of the stars our ancestors saw. This is called light pollution.
Procedure:
Obtain the equipment:
- Battery
- Decorative string lights
- Light meter (device, or mobile app for measuring - many are available for free on most phones)
- Box (like a shoebox)
- Flashlight
- Wiring
- NASA space images
- Cutting tool
Safety Tip: Use eye protection when cutting.
Begin by setting up a practice circuit that lights at least 4 decorative string lights.
- Embed the lights at the end of a shoebox (you can use a pencil to poke holes for them), and create a small viewing hole of about 2 cm at the opposite end. You will also need an opening on the side, about halfway between the two ends, that you can open and close to introduce interfering light to use the light meter and for Step 4.
- Set the box on the table. Attach the battery and make sure you can see the lights. Imagine they are stars. Can you see them all?
- Use a light meter to measure the light in the box. Your mobile app may give an answer in lux. The difference between lux and candela is that lux is a measure of the entire amount of light shining from an object. Lumens are a measure of luminous flux, the total amount of light emitted in all directions. The candela measures light we can see, coming directly from a source we can see, straight into our eyes. These differences are not significant in this introductory activity.
- Shine a flashlight inside your box. Does it affect how your eyes perceive the light?
- How does the lux or candela value change?
- How does the additional light affect how you see the "stars"?
Think:
- What real-world condition are you modeling in Step 4? Light pollution
- What problems does that condition cause? Our ability to see objects and stars in the sky is limited.
- Why would more light be a problem? Our ability to navigate and function at night is diminished.
- Find your own area on this animation: Web Link - NOAA Global Light Pollution . Is light pollution a problem where you live? How serious is the problem?
Constant:
The ampere (A) is the SI base unit of electric current. A strand of 150 decorative string lights only pulls .3 amps. The candela is the SI's base unit for measuring light as perceived by the human visual system. The lumen, a different unit, measures the light shining from an object.
Vocabulary:
- Accuracy: The degree to which the results of a measurement conform to the correct standard
- Precision: The quality of being exact; the reproducibility or repeatability of a measurement
Station 6: Physical Therapy
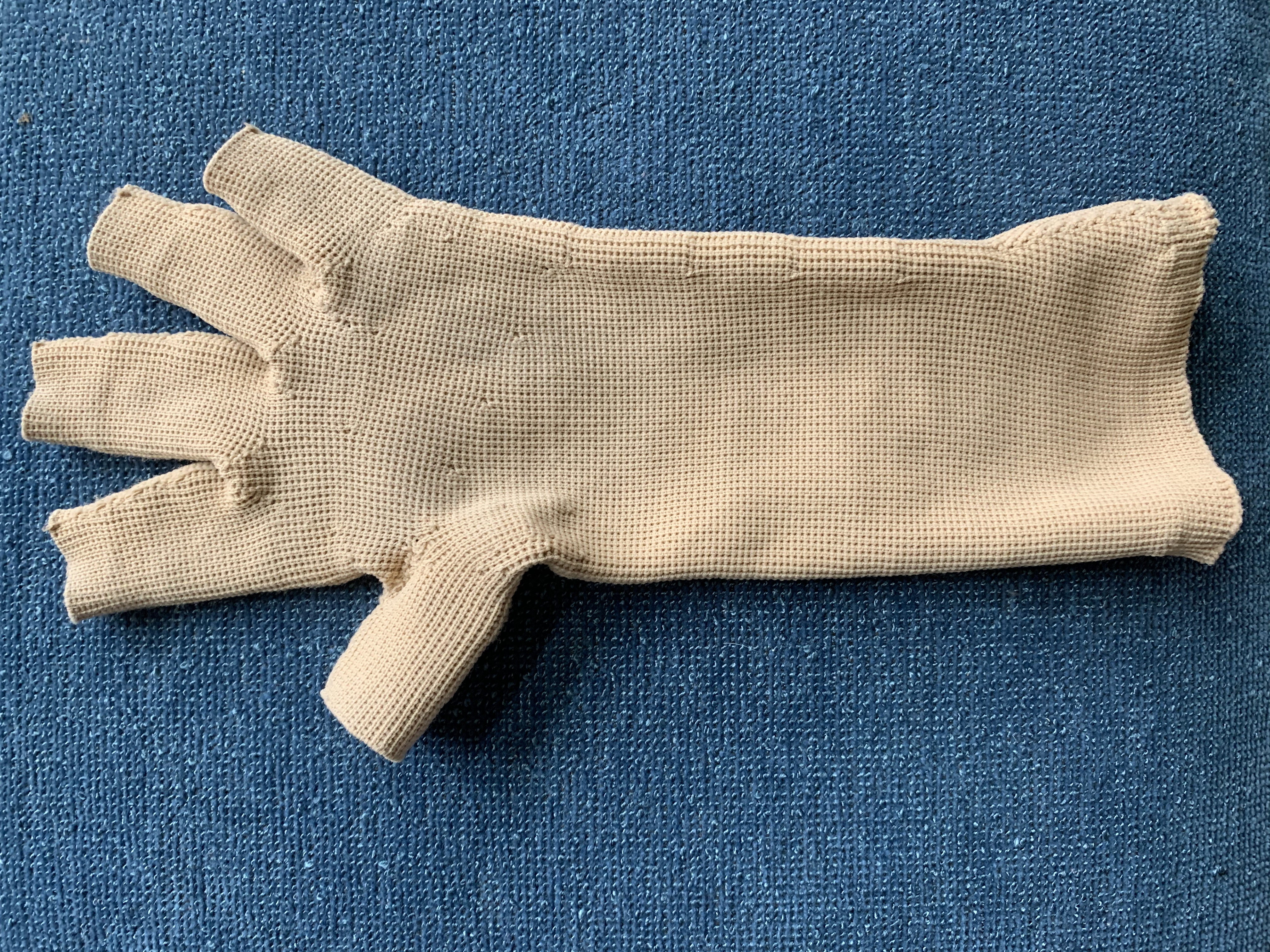
In many medical procedures, casts, gloves, or masks must be created to enable a team to apply radiation or other therapy to a precise location. When part of the body's lymph system is challenged by radiation, the therapists often create gloves to protect organs, as well as to measure stress and temperature. Making them for each client requires specialized measuring tools that are quick, individualized, and almost always disposable.
Procedure:
Use the paper measuring tool given to your group: Supporting Material - Paper measuring tool template
Cut a slit where the red line indicates, so that the graded area of the tape can slip around your finger or hand and into the slit until it is taut. Each unit is 0.4 inches, or 1.016 centimeters. Use the tool to develop a procedure to measure a hand for a specialized medical glove to be used in therapy. Create a detailed diagram of the glove that could be transmitted to a tech for 3D printing.
The glove will affect the movement of the hand by warming it. How could you embed a temperature monitor into the prosthetic? It is important that the temperature apparatus be flexible and not interfere with motion. Add a temperature measuring capacity to your design.
Share your design with another group. Ask them to give you feedback on how your design is a) practical, and b) understandable. Use that feedback to make at least one improvement in your design. While each student's design will be different, the feedback should be incorporated into an improved design.
Finally, brainstorm ideas for a different measuring tool that could be used to make an in-ear hearing aid. It must be very small and must not move much so that it doesn't irritate sensitive skin.
Think:
- Would a more precise measuring instrument help this process?
- What are the advantages of a disposable measuring tool?
- What range of temperatures would be appropriate for this device?
Constant:
The SI unit of distance is the meter, based on c, the speed of light. c is equal to 299,792,458 meters per second. Light's speed is always constant in a vacuum, so you can define any distance by the time it would take for light to travel that far. The SI unit of temperature is the Kelvin, an absolute measure of temperature that can be compared to the Celsius degree. It uses the Boltzmann constant (kB) to relate temperature to energy.
Vocabulary:
- Accuracy: The degree to which the results of a measurement conform to the correct standard
- Precision: The quality of being exact; the reproducibility or repeatability of a measurement